| <Prev Next> |
For the purposes of this example a completely blank mixture will be taken for the starting point, though it would also be possible to start from the default asymmetric top. The data is taken from "Molecular beam electric resonance spectroscopy of the nitric oxide dimer", C. M. Western, P. R. R .Langridge-Smith, B. J. Howard, and S. E. Novick, Molecular Physics, 44, 145 (1981), http://dx.doi.org/10.1080/00268978100102341.
This should be enough for a basic simulation of the microwave
spectrum; press the simulate button ( ) and then the all button (
) and then the all button (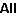 ) and you should see a simulation.
The rotational temperature is not specified in the paper, but 5
K is more likely for a van der Waals complex in a molecular beam
than the default of 300 K. Changing the plot units to MHz
("Plot", "Units" "MHz") is also more reasonable for a microwave
spectrum. Adjusting the frequency scale allows the three
microwave transitions mentioned in the paper to be identified
(at 187.5, 21224 and 22270 MHz), along with many others. The
completed data file is in nodimer.pgo
) and you should see a simulation.
The rotational temperature is not specified in the paper, but 5
K is more likely for a van der Waals complex in a molecular beam
than the default of 300 K. Changing the plot units to MHz
("Plot", "Units" "MHz") is also more reasonable for a microwave
spectrum. Adjusting the frequency scale allows the three
microwave transitions mentioned in the paper to be identified
(at 187.5, 21224 and 22270 MHz), along with many others. The
completed data file is in nodimer.pgo